Collaborators:
Amanda O’Brien, Nathan Lewis, and Sydney Miller
Introduction and Problem:
|
Through this experiment, the question of how carbon dioxide affects the pH of water was addressed. According to the Monterey Bay Aquarium Foundation, the ocean is one of the world's largest carbon sinks, absorbing more than one million metric tons of carbon dioxide every hour of the day. When carbon dioxide dissolves in water, it increases the acidity of the water, hindering the production of calcium carbonate, a compound used by many marine organisms to grow skeletons and protective shells. According to the PMEL Carbon Program, these organisms include oysters, clams, sea urchins, shallow water corals, deep sea corals, and calcareous plankton. "Between 1751 and 1994, surface ocean pH is estimated to have decreased from approximately 8.2 to 8.1, ... [which] is a nearly 26% increase in acidity" (Monterey Bay Aquarium Foundation). If this rise in acidity continues, it could lead to a devastating loss of biodiversity in marine ecosystems everywhere, as many marine organisms lose a vital compound needed for survival.
|
Corals serve as the backbone of coral reefs, which provide many marine organisms with shelter in which they can survive and reproduce. Therefore, as the acidity of the ocean continues to rise and corals continue to die, many marine organisms also suffer.
Cary, Rich. Dead Coral Reef. Digital image. Earth Times. The Earth Times, 29 Nov. 2011. Web. 2 Sept. 2014. |
Hypothesis:
If carbon dioxide is added to water, then the pH of the water will decrease because when carbon dioxide dissolves in water, it increases the hydrogen-ion concentration and therefore the pH of the solution.
Parts of the Experiment
- The control group is the 10 mL of distilled water.
- The experimental group is the 10 mL of ocean water.
- The independent variable is the type of water to which the carbon dioxide is added.
- The dependent variable is the pH of the solution measured by the color of the universal indicator after the addition of carbon dioxide.
- The controlled variables are the materials, the room temperature, the humidity of the room, the amount of water poured into each graduated cylinder, and the amount of carbon dioxide and universal indicator added to the water.
Materials:
- two test tubes
- graduated cylinder
- universal indicator
- salt water
- distilled water
- two straws
- two plastic pipets
Method:
1. Use a graduated cylinder to measure 10 mL of ocean water. Pour it into test tube #1. Add 1 mL of universal indicator. Stir and record pH in the data table.
2. Use a graduated cylinder to measure 10 mL of distilled water. Pour it into test tube #2. Add 1 mL of universal indicator. Stir and record pH in the data table.
3. Insert a straw into the ocean water sample. Slowly exhale into the water.
4. Use a stopwatch to start timing as soon as the exhaling begins and the solution starts to bubble. Stop timing as soon as the color changes. Record the data in the data table.
5. Repeat steps 3 and 4 using the sample of distilled water. Record the data in the data table.
6. Add crushed calcium carbonate to the ocean water and distilled water test tubes. Record your results.
2. Use a graduated cylinder to measure 10 mL of distilled water. Pour it into test tube #2. Add 1 mL of universal indicator. Stir and record pH in the data table.
3. Insert a straw into the ocean water sample. Slowly exhale into the water.
4. Use a stopwatch to start timing as soon as the exhaling begins and the solution starts to bubble. Stop timing as soon as the color changes. Record the data in the data table.
5. Repeat steps 3 and 4 using the sample of distilled water. Record the data in the data table.
6. Add crushed calcium carbonate to the ocean water and distilled water test tubes. Record your results.
Data:Data TablePictures |
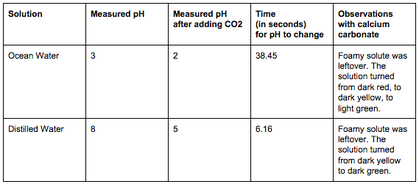
Data Analysis:The data table to the left shows the measured pH of the two solutions before and after the addition of carbon dioxide and the time that elapsed before a change in pH had occurred. The data shows that the pH of the simulated ocean water decreased by only 1 within 38.45 seconds, while the pH of the distilled water decreased by 3 within 6.16 seconds. The reason the simulated ocean water experienced a less significant and sudden change in pH is because ocean water contain a carbonate buffer system that regulates the its pH.
The data table also shows the observations made when calcium carbonate, a base, was added to both solutions. When calcium carbonate was added to the simulated ocean water, the solution turned from dark red, to dark yellow, to light green. When calcium carbonate was added to the distilled water, the solution turned from dark yellow to light green. After the addition of calcium carbonate, the two solutions were neutralized by the calcium carbonate, becoming less acidic. In addition, after the addition of calcium carbonate, both solutions had foamy, leftover solute. This leftover solute is most likely a result of an excess calcium carbonate added to each solution; fortunately, this did not skew any results since an equal amount of the compound was added to each solution. The pictures to the left show the two solutions after the addition of carbon dioxide and after the addition of calcium carbonate. The ocean water after the addition of carbon dioxide is a dark red color (picture 1). The distilled water after the addition of carbon dioxide is a dark yellow color (picture 2). The ocean water after the addition of calcium carbonate is a light green color and has a foamy, leftover solute (picture 3). The distilled water after the addition of calcium carbonate is a dark green color and also has a foamy, leftover solute (picture 4). |
Conclusions:
Conclusion Questions
1. What is the most common pH of surface ocean water?
According to the background information, the most common pH of surface ocean water is about 8.1.
2. How does your pH of ocean water compare to that reported in the background information? If there is a difference, provide possible explanations.
The measured pH of the simulated ocean water was 3, 5.1 units less than the pH of ocean water reported in the background information. The disparity between the two measurements is most likely a result of acidic chemicals leftover in the test tubes from a previous experiment. The test tubes may not have been properly cleaned, allowing any leftover chemicals to contaminate the simulated ocean water, therefore causing the solution to have an acidic pH of 3 instead of a basic pH around 8.
3. Did the distilled water and ocean water respond differently to the added carbon dioxide? Explain your results.
The distilled water and the ocean water responded differently to the addition of carbon dioxide. With the addition of carbon dioxide, the distilled water quickly became more acidic, dropping from an 8 to a 5 in pH in 6.16 seconds. On the other hand, the ocean water experienced a less significant change, slowly dropping from a 3 to a 2 in pH in 38.45 seconds. According to the Monterey Bay Aquarium Foundation, the reason that the ocean water experienced a less significant and sudden change in pH is because ocean water contains a carbonate buffer system that regulates the pH of the water. When carbon dioxide was added into the simulated ocean water, carbonate ions in the water tied up the excess hydrogen ions and produced carbonic acid, regulating the pH of the concentration. Because distilled water lacks this carbonate buffer system, its pH easily shifts from acidic to alkaline with the introduction of acids and bases.
4. Explain what happened when calcium carbonate was added to the water samples.
According to Oceana, calcium carbonate is a base; therefore, when it was added to the distilled water sample, it neutralized the acidic solution, turning the universal indicator from dark yellow to light green. When calcium carbonate was added to the simulated ocean water, it yielded similar results, turning the universal indicator from dark red, to dark yellow, and finally to dark green. In both scenarios, the solutions became less acidic with the addition of calcium carbonate. However, the distilled water solution had a final pH of about 8, while the ocean water solution had a final pH of about 5. The final pH of the distilled water solution was higher than the pH of the ocean water solution, which reiterates the important role that ocean water's carbonate buffer plays in regulating the acidity and alkalinity of the water.
5. Do you feel the [straw] experiment is a valid model for ocean absorption of carbon dioxide? Explain your answer.
The straw experiment, if conducted correctly, is a valid model of the ocean's absorption of carbon dioxide. The procedures clearly state to insert the straw into the simulated ocean water and exhale. This allows for easy absorption of carbon dioxide by the water, whereas only using the straw to exhale on the surface of the water would lead to less carbon dioxide absorbed by the water. It is also evident that the straw experiment accurately modeled the ocean's absorption of carbon dioxide because there was a change in the water's pH after air had been exhaled into the water using a straw. Because dissolved carbon dioxide decreases the pH of water, the ocean water's change in pH further substantiates the validity of the experiment.
According to the background information, the most common pH of surface ocean water is about 8.1.
2. How does your pH of ocean water compare to that reported in the background information? If there is a difference, provide possible explanations.
The measured pH of the simulated ocean water was 3, 5.1 units less than the pH of ocean water reported in the background information. The disparity between the two measurements is most likely a result of acidic chemicals leftover in the test tubes from a previous experiment. The test tubes may not have been properly cleaned, allowing any leftover chemicals to contaminate the simulated ocean water, therefore causing the solution to have an acidic pH of 3 instead of a basic pH around 8.
3. Did the distilled water and ocean water respond differently to the added carbon dioxide? Explain your results.
The distilled water and the ocean water responded differently to the addition of carbon dioxide. With the addition of carbon dioxide, the distilled water quickly became more acidic, dropping from an 8 to a 5 in pH in 6.16 seconds. On the other hand, the ocean water experienced a less significant change, slowly dropping from a 3 to a 2 in pH in 38.45 seconds. According to the Monterey Bay Aquarium Foundation, the reason that the ocean water experienced a less significant and sudden change in pH is because ocean water contains a carbonate buffer system that regulates the pH of the water. When carbon dioxide was added into the simulated ocean water, carbonate ions in the water tied up the excess hydrogen ions and produced carbonic acid, regulating the pH of the concentration. Because distilled water lacks this carbonate buffer system, its pH easily shifts from acidic to alkaline with the introduction of acids and bases.
4. Explain what happened when calcium carbonate was added to the water samples.
According to Oceana, calcium carbonate is a base; therefore, when it was added to the distilled water sample, it neutralized the acidic solution, turning the universal indicator from dark yellow to light green. When calcium carbonate was added to the simulated ocean water, it yielded similar results, turning the universal indicator from dark red, to dark yellow, and finally to dark green. In both scenarios, the solutions became less acidic with the addition of calcium carbonate. However, the distilled water solution had a final pH of about 8, while the ocean water solution had a final pH of about 5. The final pH of the distilled water solution was higher than the pH of the ocean water solution, which reiterates the important role that ocean water's carbonate buffer plays in regulating the acidity and alkalinity of the water.
5. Do you feel the [straw] experiment is a valid model for ocean absorption of carbon dioxide? Explain your answer.
The straw experiment, if conducted correctly, is a valid model of the ocean's absorption of carbon dioxide. The procedures clearly state to insert the straw into the simulated ocean water and exhale. This allows for easy absorption of carbon dioxide by the water, whereas only using the straw to exhale on the surface of the water would lead to less carbon dioxide absorbed by the water. It is also evident that the straw experiment accurately modeled the ocean's absorption of carbon dioxide because there was a change in the water's pH after air had been exhaled into the water using a straw. Because dissolved carbon dioxide decreases the pH of water, the ocean water's change in pH further substantiates the validity of the experiment.
Conclusion
The results of the experiment support the hypothesis that if carbon dioxide is added to water, then the pH of the water will decrease. According to the Monterey Bay Aquarium Foundation, this is because carbon dioxide reacts with water to form carbonic acid, which dissociates into a hydrogen ion and a bicarbonate ion. In the experiment, the water's pH decreased from a 3 to a 2 within 38.45 seconds after the addition of carbon dioxide to the simulated ocean water. Within 6.16 seconds after the addition of carbon dioxide to the distilled water, the water's pH decreased from an 8 to a 5. In both scenarios, the pH of the water decreased because the carbon dioxide dissociated into a hydrogen ion and a bicarbonate ion after it was added to the water.
Although the procedure was followed, there may have been a possible skew in the data recorded in the experiment, specifically in the initial measured pH of the simulated ocean water. According to the Monterey Bay Aquarium Foundation, the most common pH of surface ocean water is about 8.1; however, the measured pH of the simulated ocean water was 3. The skewed measurement is most likely a result of acidic chemicals leftover in the test tubes from a previous experiment. The test tubes may not have been properly cleaned, allowing any leftover chemicals to contaminate the simulated ocean water and cause the solution to have an acidic pH of 3 instead of a basic pH around 8.1.
Finally, the results justify how large amounts of carbon dioxide emissions from the burning of fossil fuels lead to ocean acidification. The experiment served as a small-scale yet valid model of carbon dioxide's affects on the ocean's pH. By inserting the straw into the simulated ocean water and exhaling, the absorption of carbon dioxide by the ocean was simulated, which resulted in a slight decrease in the water's pH. Since the simulated ocean water had a Knudsen salinity of 35.00 ppt, the most common Knudsen salinity of ocean water, the results of the small-scale experiment can be extrapolated to a large-scale situation, and it can be concluded that carbon dioxide leads to ocean acidification. "The 2013 average annual concentration of CO2 in the atmosphere was 396.48 parts per million" (Annual Data: Atmospheric Carbon Dioxide), and according to the Monterey Bay Aquarium, the ocean absorbs more than one million metric tons of carbon dioxide every hour. If the levels of carbon dioxide emissions do not decrease, they could lead to a drastic loss of biodiversity in marine ecosystems.
Although the procedure was followed, there may have been a possible skew in the data recorded in the experiment, specifically in the initial measured pH of the simulated ocean water. According to the Monterey Bay Aquarium Foundation, the most common pH of surface ocean water is about 8.1; however, the measured pH of the simulated ocean water was 3. The skewed measurement is most likely a result of acidic chemicals leftover in the test tubes from a previous experiment. The test tubes may not have been properly cleaned, allowing any leftover chemicals to contaminate the simulated ocean water and cause the solution to have an acidic pH of 3 instead of a basic pH around 8.1.
Finally, the results justify how large amounts of carbon dioxide emissions from the burning of fossil fuels lead to ocean acidification. The experiment served as a small-scale yet valid model of carbon dioxide's affects on the ocean's pH. By inserting the straw into the simulated ocean water and exhaling, the absorption of carbon dioxide by the ocean was simulated, which resulted in a slight decrease in the water's pH. Since the simulated ocean water had a Knudsen salinity of 35.00 ppt, the most common Knudsen salinity of ocean water, the results of the small-scale experiment can be extrapolated to a large-scale situation, and it can be concluded that carbon dioxide leads to ocean acidification. "The 2013 average annual concentration of CO2 in the atmosphere was 396.48 parts per million" (Annual Data: Atmospheric Carbon Dioxide), and according to the Monterey Bay Aquarium, the ocean absorbs more than one million metric tons of carbon dioxide every hour. If the levels of carbon dioxide emissions do not decrease, they could lead to a drastic loss of biodiversity in marine ecosystems.
Citations:
Cary, Rich. Dead Coral Reef. Digital image. Earth Times. The Earth Times, 29 Nov. 2011. Web. 2 Sept. 2014.
The Power of PH: Changing Ocean Chemistry. N.p.: Monterey Bay Aquarium Foundation, 2010. PDF.
"What Is Ocean Acidification?" Oceana. N.p., 2012. Web. 30 Aug. 2014. <http://oceana.org/en/our-work/climate-energy/ocean-acidification/learn-act/what-is-ocean-acidification>.
"What Is Ocean Acidification?." PMEL Carbon Program. National Oceanic and Atmospheric Administration, n.d. Web. 2 Sept. 2014. <http://www.pmel.noaa.gov/co2/story/Ocean+Acidification>.
The Power of PH: Changing Ocean Chemistry. N.p.: Monterey Bay Aquarium Foundation, 2010. PDF.
"What Is Ocean Acidification?" Oceana. N.p., 2012. Web. 30 Aug. 2014. <http://oceana.org/en/our-work/climate-energy/ocean-acidification/learn-act/what-is-ocean-acidification>.
"What Is Ocean Acidification?." PMEL Carbon Program. National Oceanic and Atmospheric Administration, n.d. Web. 2 Sept. 2014. <http://www.pmel.noaa.gov/co2/story/Ocean+Acidification>.