Collaborators:
Joshua Arteta, Carly Sullivan, and Nicole Sullivan
Introduction and Problem:
The LD-50 of a compound, "the individual dose [of that compound] required to kill fifty percent of a population of [organisms]" (Environmental Protection Agency), is calculated in many fields of science, especially pharmacology, in order to assess the risks of certain chemical compounds. Scientists calculate the LD-50 of a compound using a dose-response experiment in which different populations of test organisms are exposed to various doses of that compound. The data can then be used to manage the risks of that compound; for example, according to the background information, it is suspected that the run-off of salt used to deice highways may affect the growth of nearby vegetation and aquatic plants. By conducting a dose-response experiment on radish seeds using different salt concentrations, the concentration of salt (mg/L) required to kill fifty percent of a radish seed population will be calculated, ultimately assessing the risk of deicing roads with salt.
Hypothesis:
If radish seeds are exposed to 12.0 mg/L of salt, then fifty percent of the population of seeds will die, and the seeds that do germinate will have radicles that are significantly shorter than those of the control group.
Parts of the Experiment:
- The independent variable is the concentration of salt (mg/L) that is added to the radish seeds.
- The dependent variables are the number of seeds that germinate and the average length of each radicle.
- The controlled variables are the materials, the amount of water each plant receives, and the amount of light each plant is exposed to, and the number of radish seeds per bag.
- The control group consists of the seeds exposed to distilled water.
- The experimental group consists of the seeds exposed to various concentrations of salt.
Materials:
- 6 test tubes
- test tube rack
- pipette
- distilled water
- concentrated salt solution (12.0 grams of table salt per 1 L of distilled water)
- sparkling radishes
- 6-90 mm petri dish
- 3 pieces of unbleached paper towels
- 6 plastic bags
- metric ruler
Method:
Part 1: Preparing Serial Dilutions of a Salt Solution
1. Set up 6 test tubes in a test tube rack and label the tubes with the following salt
concentrations: 12.0 g/L, 6.0 g/L, 3.0 g/L, 1.5 g/L, 0.75 g/L and Control. See Table 1
Salt Solutions.
2. Add 10 mL of distilled water to test tubes #2- 6.
3. Measure 20 mL of the concentrated solution (12.0 g/L) and pour into test tube #1.
4. Transfer 10 mL of salt solution from test tube #1 to test tube #2.
5. Gently swirl test tube #2 to mix the salt solution.
6. Repeat steps 4 and 5 for test tubes #3-5 measuring 10 mL each time. DO NOT add
any salt solution to test tube #6.
7. Measure 10 mL of distilled water into test tube #6 to serve as the control. The control
will indicate whether or not your seeds are viable (capable of growing or
developing).
8. Unless you will be using the solutions right away, cover them tightly with plastic
wrap to prevent water loss through evaporation.
Part 2: Setting up a Dose-Response Experiment
1. Obtain six plastic bags. Label each dish according to the concentration of salt solution
to be tested. See Table 1. Salt Solution Concentrations.
2. Fold a half of sheet of paper towel or coffee filter into quarters. Cut it out so that it
fits into the bottom of the plastic bag.
3. Measure 6 ml of salt solution and pour onto the paper towel in the appropriate petri
dish. In the control dish, add 5 mL of distilled water. The purpose of a control is to
identify how well the seeds will grow without any salt.
4. Add 10 radish seeds to each plastic bag. Space the seeds out evenly on the paper
towel so that they do not touch each other or the sides of the dish.
5. Place the dishes in a plastic bag and seal it to retain moisture. Label your group/s
name on the outside of the bag.
6. Incubate the seeds in a dark place at a constant temperature for a couple of days.
T. Inspect radish seeds during incubation period. Record any observations. If the paper
seems dry, add a 1 or 2 more millimeters of the appropriate salt solution or distilled
water (control).
Part 3: Collecting Data and Plotting Results
1. Remove the lid of the control dish. Count the number of seeds that germinated
(sprouted). Calculate the percentage of seeds that germinated and record in Table 2.
Radish Seed Results.
2. To measure the length of the radicle (embryonic root), carefully remove the
germinating radish seed from the paper towel in one piece. The radicle may be
growing into the layers of towel and can break if you pull too hard.
3. Measure the length of the radicle for each of the germinating radish seeds to the
nearest millimeter (mm). Look carefully at each sprout to make sure you are
measuring just the root, not the shoot as well. In the picture below, you would
measure just the part between the two arrows, not the shoot and cotyledons to the
left. Record data in Table 2. Radish Seed Results.
4. Repeat steps 1-3 for each plastic bag.
5. For each treatment, calculate the mean (arithmetic average) radicle length for each
salt solution. Add the total radicle lengths for each salt solution and divide by the total number of seeds that germinated. Record data in column labeled "Mean Radicle Length (mm)" in Table 2. Radish Seed Results.
1. Set up 6 test tubes in a test tube rack and label the tubes with the following salt
concentrations: 12.0 g/L, 6.0 g/L, 3.0 g/L, 1.5 g/L, 0.75 g/L and Control. See Table 1
Salt Solutions.
2. Add 10 mL of distilled water to test tubes #2- 6.
3. Measure 20 mL of the concentrated solution (12.0 g/L) and pour into test tube #1.
4. Transfer 10 mL of salt solution from test tube #1 to test tube #2.
5. Gently swirl test tube #2 to mix the salt solution.
6. Repeat steps 4 and 5 for test tubes #3-5 measuring 10 mL each time. DO NOT add
any salt solution to test tube #6.
7. Measure 10 mL of distilled water into test tube #6 to serve as the control. The control
will indicate whether or not your seeds are viable (capable of growing or
developing).
8. Unless you will be using the solutions right away, cover them tightly with plastic
wrap to prevent water loss through evaporation.
Part 2: Setting up a Dose-Response Experiment
1. Obtain six plastic bags. Label each dish according to the concentration of salt solution
to be tested. See Table 1. Salt Solution Concentrations.
2. Fold a half of sheet of paper towel or coffee filter into quarters. Cut it out so that it
fits into the bottom of the plastic bag.
3. Measure 6 ml of salt solution and pour onto the paper towel in the appropriate petri
dish. In the control dish, add 5 mL of distilled water. The purpose of a control is to
identify how well the seeds will grow without any salt.
4. Add 10 radish seeds to each plastic bag. Space the seeds out evenly on the paper
towel so that they do not touch each other or the sides of the dish.
5. Place the dishes in a plastic bag and seal it to retain moisture. Label your group/s
name on the outside of the bag.
6. Incubate the seeds in a dark place at a constant temperature for a couple of days.
T. Inspect radish seeds during incubation period. Record any observations. If the paper
seems dry, add a 1 or 2 more millimeters of the appropriate salt solution or distilled
water (control).
Part 3: Collecting Data and Plotting Results
1. Remove the lid of the control dish. Count the number of seeds that germinated
(sprouted). Calculate the percentage of seeds that germinated and record in Table 2.
Radish Seed Results.
2. To measure the length of the radicle (embryonic root), carefully remove the
germinating radish seed from the paper towel in one piece. The radicle may be
growing into the layers of towel and can break if you pull too hard.
3. Measure the length of the radicle for each of the germinating radish seeds to the
nearest millimeter (mm). Look carefully at each sprout to make sure you are
measuring just the root, not the shoot as well. In the picture below, you would
measure just the part between the two arrows, not the shoot and cotyledons to the
left. Record data in Table 2. Radish Seed Results.
4. Repeat steps 1-3 for each plastic bag.
5. For each treatment, calculate the mean (arithmetic average) radicle length for each
salt solution. Add the total radicle lengths for each salt solution and divide by the total number of seeds that germinated. Record data in column labeled "Mean Radicle Length (mm)" in Table 2. Radish Seed Results.
Data Analysis:
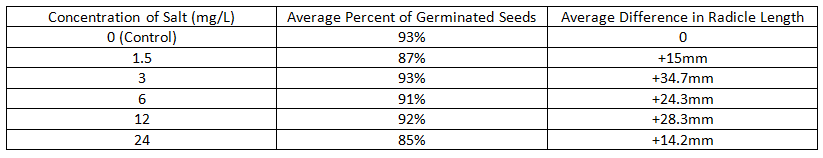
Data Table
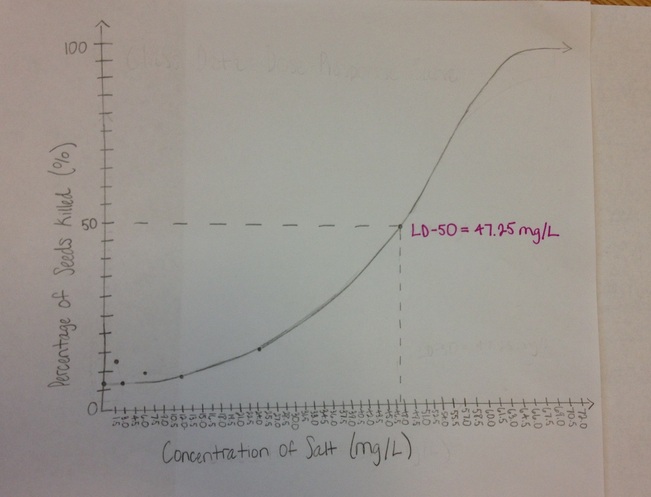
Dose-Response Curve
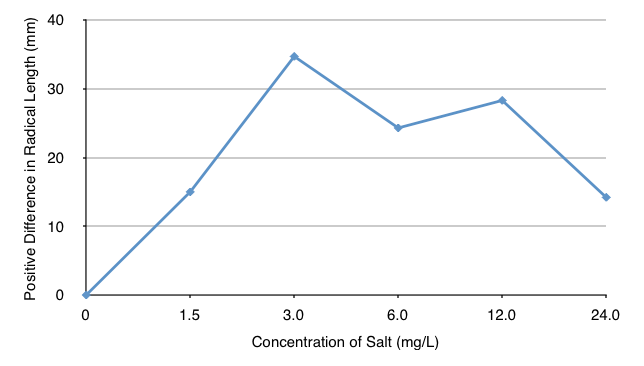
Difference in Radicle Length Graph
Pictures |
AnalysisAfter the class data had been organized using a table, some basic trends in the data were discovered: On average, radish seeds exposed to 12 mg/L (picture 4) and 24 mg/L of salt (picture 5) both tended to have short radicle lengths. Also, exposing radish seeds to 3 mg/L of salt led to the germination of 93% of the seeds and radicle lengths 34.7 mm greater than those of the control group, while exposing seeds to 24 mg/L of salt led to the germination of 85% of the seeds and radicle lengths 14.2 mm greater than those of the control group. However, there may have been a possible skew in the data of the experiment: many of the radish seeds exposed to 24mg/L of salt still germinated and had radicles longer than those of the control group.
In addition, the radish seeds exposed to 12 mg/L and 24 mg/L of salt had bulbs that were more yellow than those of the seeds exposed to 0 mg/L (picture 1), 1.5 mg/L (picture 2), and 3 mg/L of salt, which had dark green bulbs. The disparity between the colors of the bulbs of the seeds exposed to high concentrations and the seeds exposed to low concentrations of salt is not arbitrary; instead, there is a strong correlation between the salt concentration that seeds are exposed to, and the color of their bulbs. After extrapolating the class data onto a dose-response curve, the LD-50 of salt on radish seeds was predicted to be 47.25 mg/L, meaning that if a population of radish seeds were exposed to 47.25 mg/L of salt, half of the population would not germinate. |
Conclusions:
The results of this experiment contradicted the hypothesis that fifty percent of radish seed population exposed to 12.0 mg/L of salt would die, and that the seeds that germinated would have shorter radicle lengths than those of the control group. According to the class data, the average percent of radish seeds that germinated after being exposed to each concentration of salt did not reach 50%; the lowest percentage of seeds that germinated, 85%, resulted from exposing them to the highest concentration of salt, 24 mg/L. Therefore, it can be predicted that the LD-50 of salt on radish seeds must be greater than 24 mg/L, and after extrapolating the data onto a dose-response curve, the LD-50 of salt on radish seeds was predicted to be 47.25 mg/L.
In addition, the concentration of salt that germinated the highest percentage of seeds with the longest radicles was 3 mg/L. The percentage of seeds that germinated was also the same as that of the control group; therefore, it can be predicted that concentrations of salt less than or equal to 3 mg/L of salt are nontoxic to radish seeds.
Finally, the radish seeds with the most yellow bulbs were those that were exposed to 6 g/L of salt or higher (pictures 3, 4, and 5). Although the number of these radish seeds that germinated and the average length of their radicles remained high, their yellow bulbs may indicate that seeds exposed to salt concentrations of 6 g/L or higher may still germinate and grow long radicles, but will grow less healthy bulbs.
However, there may have been a skew in the data collected: a strong correlation did not exist between the independent variable (the concentration of salt the seeds are exposed to) and the dependent variable (the number of seeds that germinate and the average length of each radicle). On average, exposing radish seeds to 1.5 mg/L of salt led to the germination of 87% of the seeds and radicle lengths 15 mm greater than the control group, while exposing radish seeds to 12 mg/L of salt led to the germination of 92% of the seeds and radicle lengths 14.2 mm greater than the control group. The radish seeds exposed to 10.5 mg/L more of salt led to the germination of 5% more of the seeds and insignificantly shorter radicle lengths. This skew in the data may have been a result of some groups incorrectly labeling their bags of seeds with the wrong salt concentrations. As a result, the average percentages of seeds that germinated and the average length of each radicle were skewed.
Although the LD-50 of salt on radish seeds could not be calculated solely based on the group data, after extrapolating the data onto a dose-response curve, the LD-50 was predicted to be 47.25 mg/L. "Deicing salt is usually refined rock salt consisting of about 98.5 percent sodium chloride, 1.2 percent calcium sulfate, 0.1 percent magnesium chloride, and 0.2 percent rock" (Perry, Leonard). Because the concentration of salt used to deice roads falls above the LD-50 of salt on radish seeds, it can be predicted that these levels are high enough to severely harm nearby vegetation and aquatic plant populations; therefore, the risk can be managed in order to prevent severe harm to the environment: [Roadsides can be protected] by constructing burlap or durable plastic screens to shield them from [run-off of salt used to deice roads during the winter]" (Lerner, Rosie).
In addition, the concentration of salt that germinated the highest percentage of seeds with the longest radicles was 3 mg/L. The percentage of seeds that germinated was also the same as that of the control group; therefore, it can be predicted that concentrations of salt less than or equal to 3 mg/L of salt are nontoxic to radish seeds.
Finally, the radish seeds with the most yellow bulbs were those that were exposed to 6 g/L of salt or higher (pictures 3, 4, and 5). Although the number of these radish seeds that germinated and the average length of their radicles remained high, their yellow bulbs may indicate that seeds exposed to salt concentrations of 6 g/L or higher may still germinate and grow long radicles, but will grow less healthy bulbs.
However, there may have been a skew in the data collected: a strong correlation did not exist between the independent variable (the concentration of salt the seeds are exposed to) and the dependent variable (the number of seeds that germinate and the average length of each radicle). On average, exposing radish seeds to 1.5 mg/L of salt led to the germination of 87% of the seeds and radicle lengths 15 mm greater than the control group, while exposing radish seeds to 12 mg/L of salt led to the germination of 92% of the seeds and radicle lengths 14.2 mm greater than the control group. The radish seeds exposed to 10.5 mg/L more of salt led to the germination of 5% more of the seeds and insignificantly shorter radicle lengths. This skew in the data may have been a result of some groups incorrectly labeling their bags of seeds with the wrong salt concentrations. As a result, the average percentages of seeds that germinated and the average length of each radicle were skewed.
Although the LD-50 of salt on radish seeds could not be calculated solely based on the group data, after extrapolating the data onto a dose-response curve, the LD-50 was predicted to be 47.25 mg/L. "Deicing salt is usually refined rock salt consisting of about 98.5 percent sodium chloride, 1.2 percent calcium sulfate, 0.1 percent magnesium chloride, and 0.2 percent rock" (Perry, Leonard). Because the concentration of salt used to deice roads falls above the LD-50 of salt on radish seeds, it can be predicted that these levels are high enough to severely harm nearby vegetation and aquatic plant populations; therefore, the risk can be managed in order to prevent severe harm to the environment: [Roadsides can be protected] by constructing burlap or durable plastic screens to shield them from [run-off of salt used to deice roads during the winter]" (Lerner, Rosie).
Citations:
Lerner, Rosie. "Purdue University Consumer Horticulture." Deicing Salts Harmful to Plants. N.p., n.d. Web. 17 Sept. 2014. <http://www.hort.purdue.edu/ext/deicingsalts.html>.
"Lethal Dosage (LD50) Values." EPA. Environmental Protection Agency, 27 June 2012. Web. 13 Sept. 2014. <http://www.epa.gov/oecaagct/ag101/pestlethal.html>.
Perry, Leonard. "Salt Damage to Plants." University of Vermont. N.p., n.d. Web. 13 Sept. 2014. <http://www.uvm.edu/pss/ppp/articles/salt1.htm>.
"Lethal Dosage (LD50) Values." EPA. Environmental Protection Agency, 27 June 2012. Web. 13 Sept. 2014. <http://www.epa.gov/oecaagct/ag101/pestlethal.html>.
Perry, Leonard. "Salt Damage to Plants." University of Vermont. N.p., n.d. Web. 13 Sept. 2014. <http://www.uvm.edu/pss/ppp/articles/salt1.htm>.